Concentration
Feedstock is normally concentrated prior to introduction into the spray dryer. The concentration stage increases the solids content thereby reducing the amount of liquid that must be evaporated in the spray dryer.
Atomization
Spray dryers are characterized by the atomization of the feedstock and the contacting of the spray with heated air. The atomization stage is designed to create the optimum conditions for evaporation and to lead to a dried product having the desired characteristics. Nozzles and rotary atomizers are used to form sprays. Dryers can range from just one nozzle to having over fifty.
Droplet-air contact
The central element of a spray dryer is the spray dry chamber. In the chamber, atomized liquid is brought into contact with hot gas (usually air, at a vacuum), resulting in the evaporation of 95%+ of the water contained in the droplets in a matter of a few seconds. The way in which the spray makes contact with the air in the dryer influences the behavior of the droplet during the drying phase. This has a direct bearing on the properties of the dried product. The type of contact between the spray and the air is determined by the position of the atomizer relative to the air inlet. Nozzle headers are usually located at the top of the dryer and spray down.
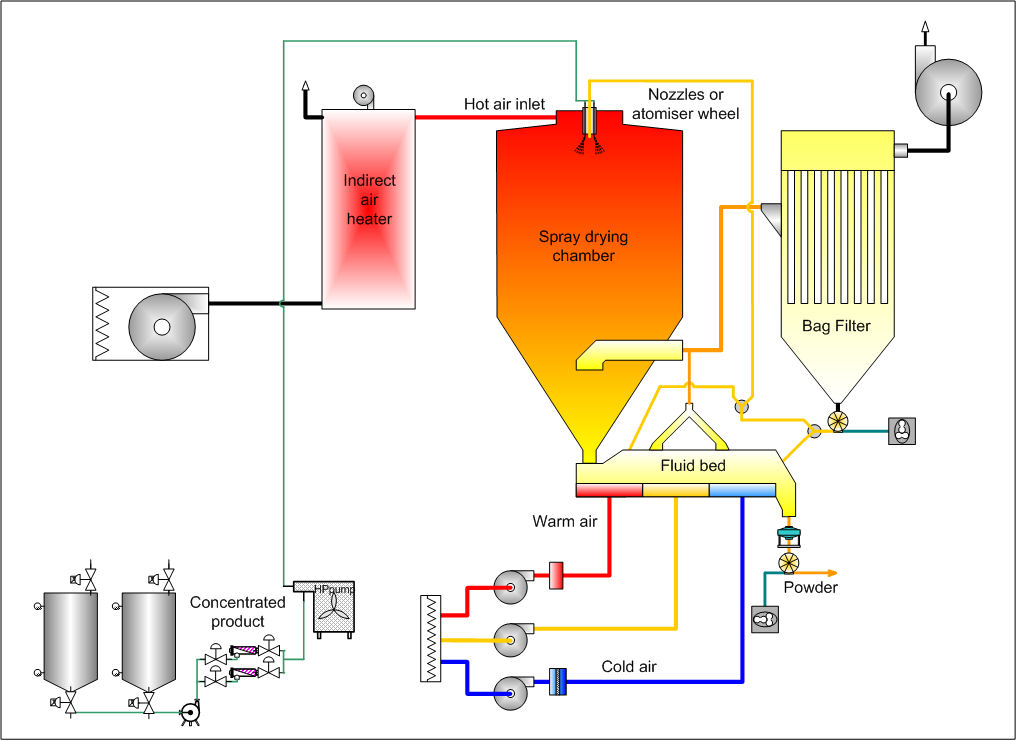
WPE-BV
Droplet drying
First stage
Moisture evaporation takes place in two stages. During the first stage, the temperature in the saturated air at the surface of the droplet is approximately equal to the wet-bulb temperature of the drying air. There is sufficient moisture in the drop to replace the liquid evaporated at the surface and evaporation takes place at a relatively constant rate.
Second stage
The second stage begins when there is no longer enough moisture to maintain saturated conditions at the droplet surface, causing a dried shell to form at the surface. Evaporation then depends on the diffusion of moisture through the shell, which is increasing in thickness. The rate of evaporation falls rapidly during the second phase. Different products have differing evaporation and particle-forming characteristics. Some expand, others contract, fracture or disintegrate.
The resulting particles may be relatively uniform hollow spheres, or porous and irregularly shaped.
Separation
Following completion of drying, the particles of product must be separated from the drying air. Primary separation is accomplished by the particles simply falling to the bottom of the chamber. A small fraction of the particles remain entrained with the air and must be recovered in separation equipment.
Final separation stage
Cyclones, bag filters, and electrostatic precipitators may be used for the final separation stage. Wet scrubbers are then often used to purify and cool the air so that it can be released to atmosphere.
